The primary way of preparing alkenes is through elimination reactions of alkyl halides and alcohols. The key difference between these two substrates is that the elimination of alkyl halides follows the E2 mechanism, while alcohols are dehydrated to alkenes via an E1 mechanism.
Let’s start with the synthesis of alkenes from alkyl halides.
Alkenes from Alkyl Halides
We can prepare an alkene from an alkyl halide using a strong base such as NaOH or NaOEt, or a bulky base such as potassium tert-butoxide to obtain the less substituted (Hofmann) alkene.
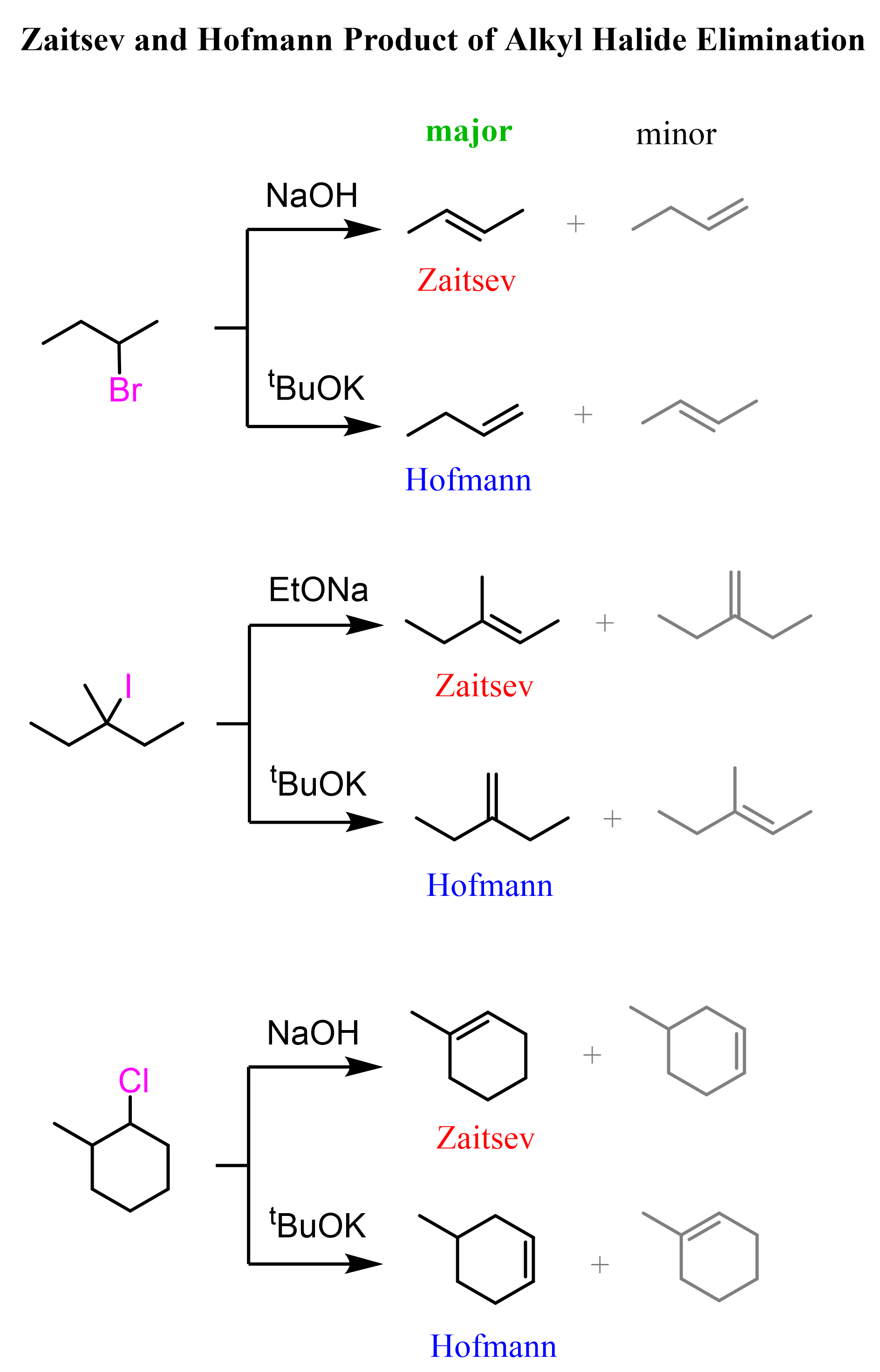
Once again, notice that we control the regiochemistry of the elimination reaction by using either a sterically unhindered base to obtain the Zaitsev product, or a bulky base to obtain the Hofmann product.
The E1 Elimination of Alkyl Halides
Alkyl halides can also be converted to alkenes by an E1 mechanism if a weak base, such as water or an alcohol, is used.

Remember that the SN1 reaction is always in competition with the E1 reaction; therefore, to favor the elimination path, heat is most often used.
Check this article on comparing the SN1 and E1 reactions for more details, including the effect of temperature on the substitution and elimination reactions.
Alknes from Dehydration of Alcohols
As mentioned at the beginning, another way of preparing alkenes is the dehydration of alcohols. The conversion is often carried out by using a concentrated solution of sulfuric acid, which first protonates the OH groups, converting it into an excellent leaving group. Once water is lost, a carbocation intermediate is formed, which is attacked by water, forming the final product alkene.

Using concentrated acids has its drawbacks, as not every molecule or functional group survives these conditions.
Another issue here is the formation of a carbocation, which always comes with the risk of a rearrangement. Therefore, the alternative approach is to convert the alcohol to a mesylate or a tosylate and react it with a strong base to promote an E2 elimination, thus excluding the possibility of a rearrangement.

A great alternative to above above-mentioned approaches is the use of POCl3 in the presence of an organic base such as pyridine. This method is regioselective, and the Zaitsev product is obtained:

Preparation of Alkenes from Alkynes
Alkenes can also be prepared by reducing the corresponding alkyne, and the stereochemistry of the resulting alkene depends on the choice of catalyst and reaction conditions.
For cis (Z) alkenes, a terminal or internal alkyne can be partially hydrogenated using Lindlar’s catalyst, which is palladium on calcium carbonate poisoned with lead or quinoline.

For trans (E) alkenes, the alkyne can be reduced using a dissolving metal reduction, such as sodium or lithium in liquid ammonia (NH₃). This method results in a trans-alkene because the hydrogens add in an anti-addition, giving opposite sides of the former triple bond.

Alkenes from Carbonyl Compounds
This is an organic II topic, but I will add it here in case you have reached it in class. The conversion is called the Wittig reaction, which transforms carbonyl compounds, such as aldehydes and ketones, into alkenes using a phosphonium ylide (Ph₃P=CR₂).
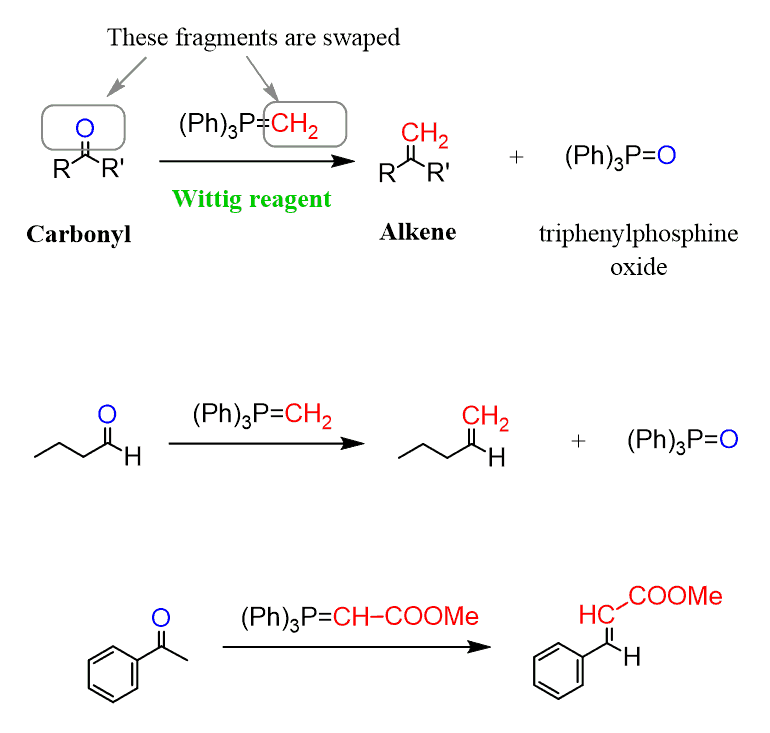
Mechanism: The ylide attacks the carbonyl carbon, forming a betaine intermediate, which then collapses to give the alkene and triphenylphosphine oxide.

This reaction is a powerful method to synthesize alkenes directly from aldehydes or ketones, especially when precise control of the double bond location is required.
Check Also
- Alkenes: Structure and Stability
- Naming Alkenes by IUPAC Nomenclature Rules
- Cis and Trans Isomers
- E and Z Alkene Configuration with Practice Problems
- Electrophilic Addition Reactions to Alkenes
- Markovnikov’s Rule
- Markovnikov’s Rule with Practice Problems
- Addition of Water to Alkenes
- Acid-Catalyzed Hydration of Alkenes with Practice Problems
- Rearrangements in Alkene Addition Reactions
- Oxymercuration-Demercuration
- Addition of Alcohols to Alkenes
- Free-Radical Addition of HBr: Anti-Markovnikov Addition
- Hydroboration-Oxidation: The Mechanism
- Hydroboration-Oxidation of Alkenes: Regiochemistry and Stereochemistry with Practice Problems
- Halogenation of Alkenes and Halohydrin Formation
- The Regiochemistry of Alkene Addition Reactions
- The Stereochemistry of Alkene Addition Reactions
- Cis product in an anti-Addition Reaction of Alkenes
- Ozonolysis of Alkenes with Practice Problems
- Syn Dihydroxylation of Alkenes with KMnO4 and OsO4
- Anti-Dihydroxylation of Alkenes with MCPBA and Other Peroxides with Practice Problems
- Oxidative Cleavage of Alkenes with KMno4 and O3
- Alkene Reactions Practice Problems
- Changing the Position of a Double Bond
- Changing the Position of a Leaving Group
- Alkenes Multi-Step Synthesis Practice Problems
- Alkene Addition Reactions Practice Quiz
- Reactions Map of Alkenes
