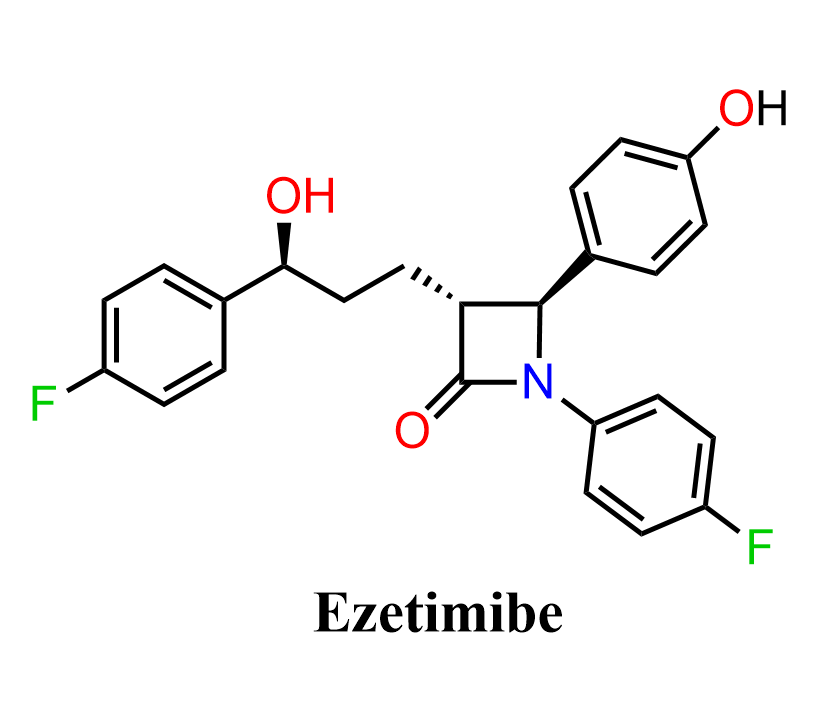
 Ezetimibe is a cholesterol absorption inhibitor used in the treatment of hyperlipidemia, which is a common, often asymptomatic condition characterized by high levels of lipids (cholesterol/triglycerides) in the blood, which can lead to plaque buildup in arteries.
Ezetimibe is a cholesterol absorption inhibitor used in the treatment of hyperlipidemia, which is a common, often asymptomatic condition characterized by high levels of lipids (cholesterol/triglycerides) in the blood, which can lead to plaque buildup in arteries.
Structurally, it is characterized by a β-lactam (azetidinone) core, substituted with aryl groups and a side chain bearing a secondary alcohol. The presence of multiple stereocenters and functional groups makes it a valuable target in synthetic organic chemistry. Its biological activity arises from inhibition of intestinal cholesterol uptake, rather than direct interaction with cholesterol biosynthesis pathways.
Because of its structure, ezetimibe is often discussed in organic chemistry in the context of β-lactam formation, stereoselective synthesis, protecting group strategies, and carbon–carbon bond-forming reactions.
In the following practice problem, we will focus on the organic synthesis of ezetimibe rather than its pharmacological properties. Specifically, we will examine how the azetidinone framework can be constructed starting from simple aromatic precursors such as phenol, using key transformations such as imine formation, ketene–imine cycloaddition, and subsequent functional group interconversions to arrive at the final molecule.
As with all examples in this section, this discussion is presented solely for educational purposes and is not intended to represent a practical or real-world method of preparation.
Practice
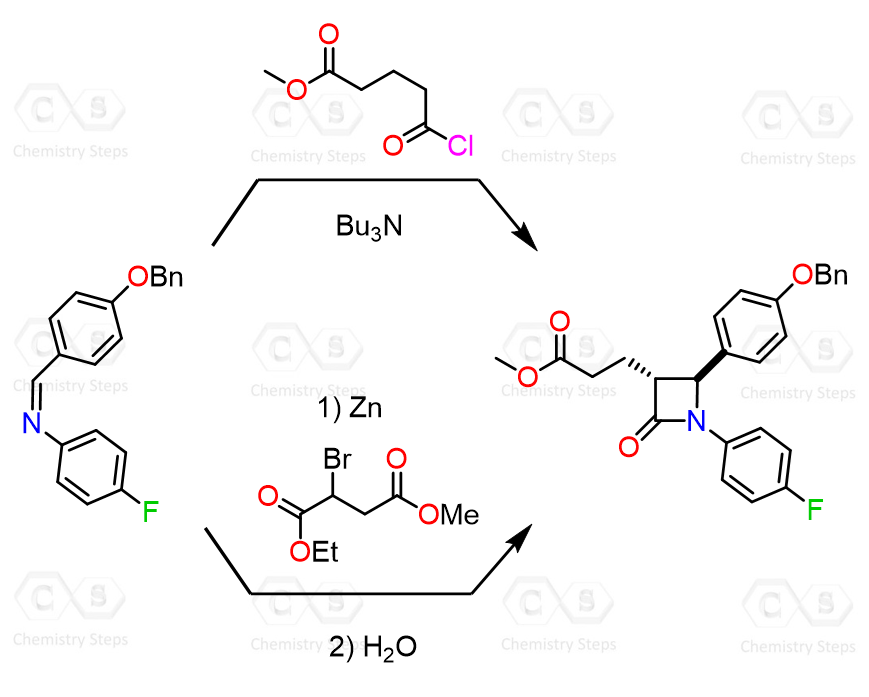
Add the missing intermediates and reagents in the following synthesis of Ezetimibe. Draw a curved arrow mechanism for the conversion 3-4, where the β-lactam ring is formed.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.