We know that esters undergo different types of condensation reactions when their enolate are formed upon reacting with strong bases. The most common ones are the Claisen, Dieckmann, and also the crossed aldol condensation, where the enolate of the ester is reacted with a different carbonyl compound, such as, for example, an aldehyde or a ketone:
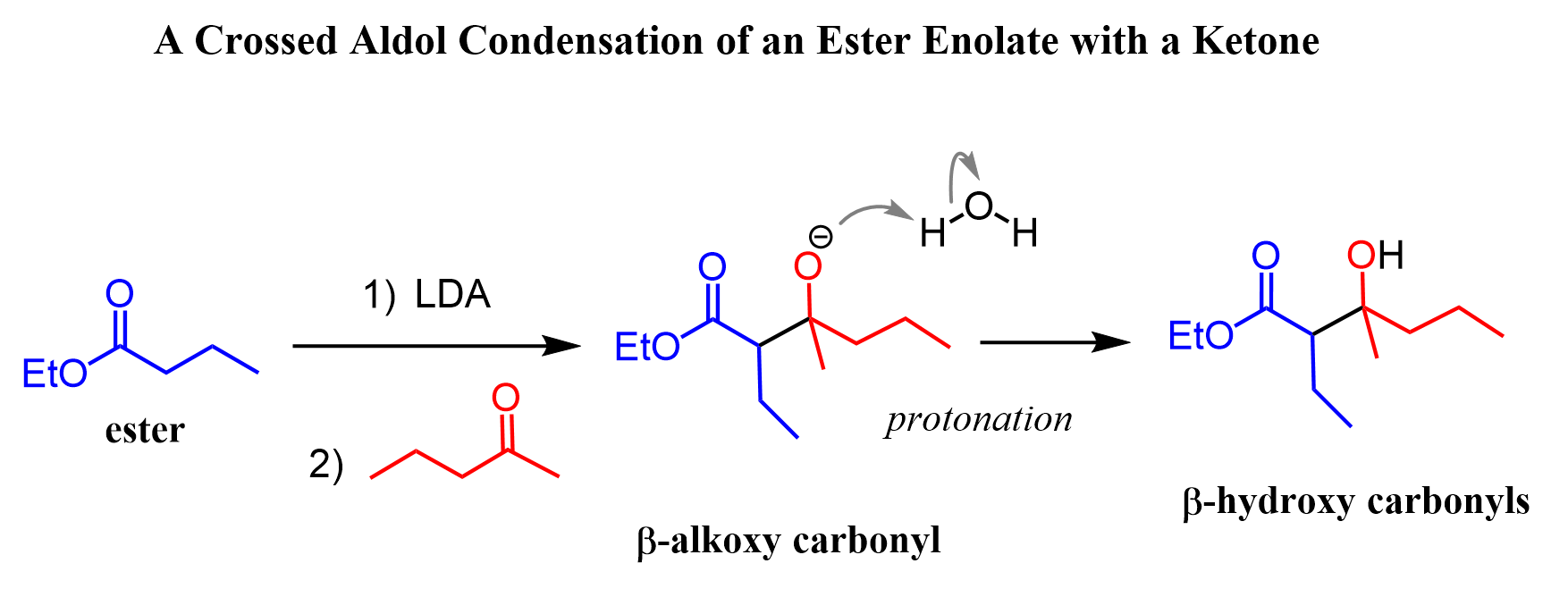
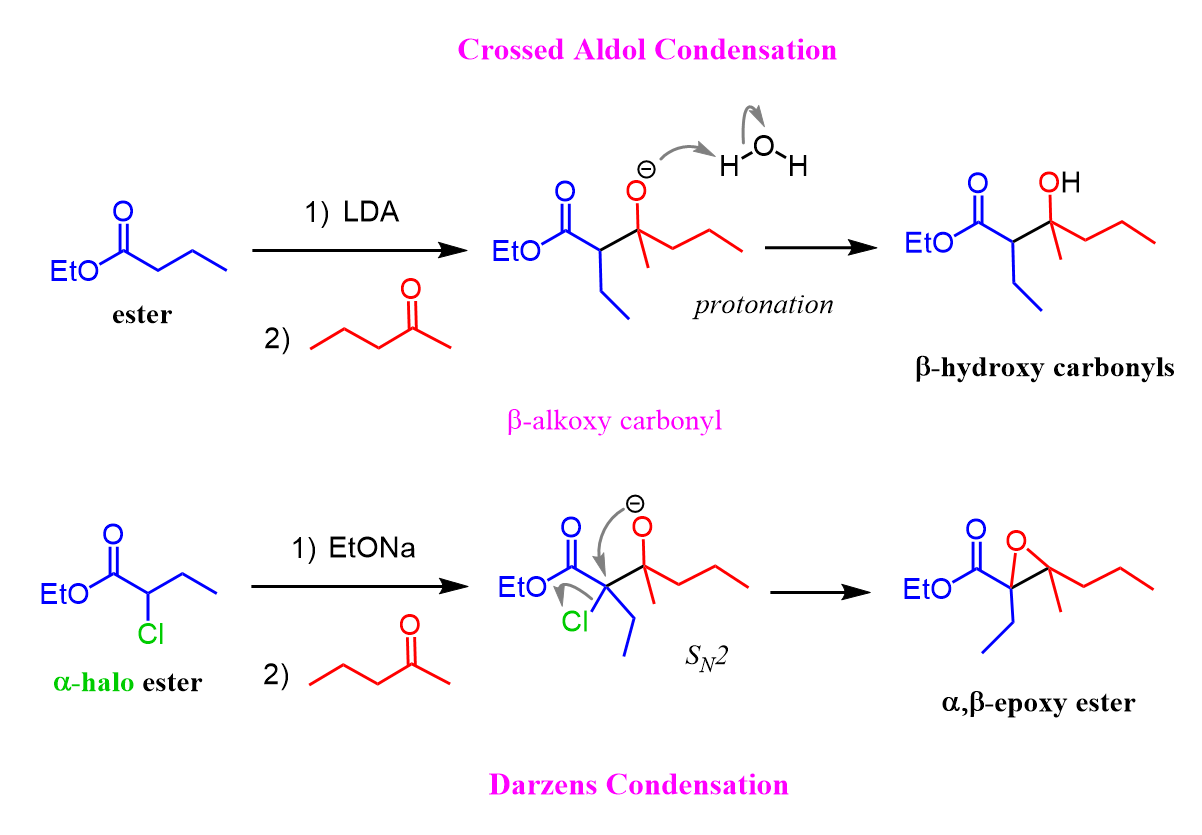
Remember, what happens is the enolate is formed and then attacks the carbonyl group to form a β-alkoxy carbonyl, which, upon protonation, is transferred to a β-hydroxy carbonyl.
Ok, so let’s now see what happens if the ester has a halogen on the α position – that is, we are making the enolate of an α-halo ester and then reacting it with a ketone or an aldehyde:
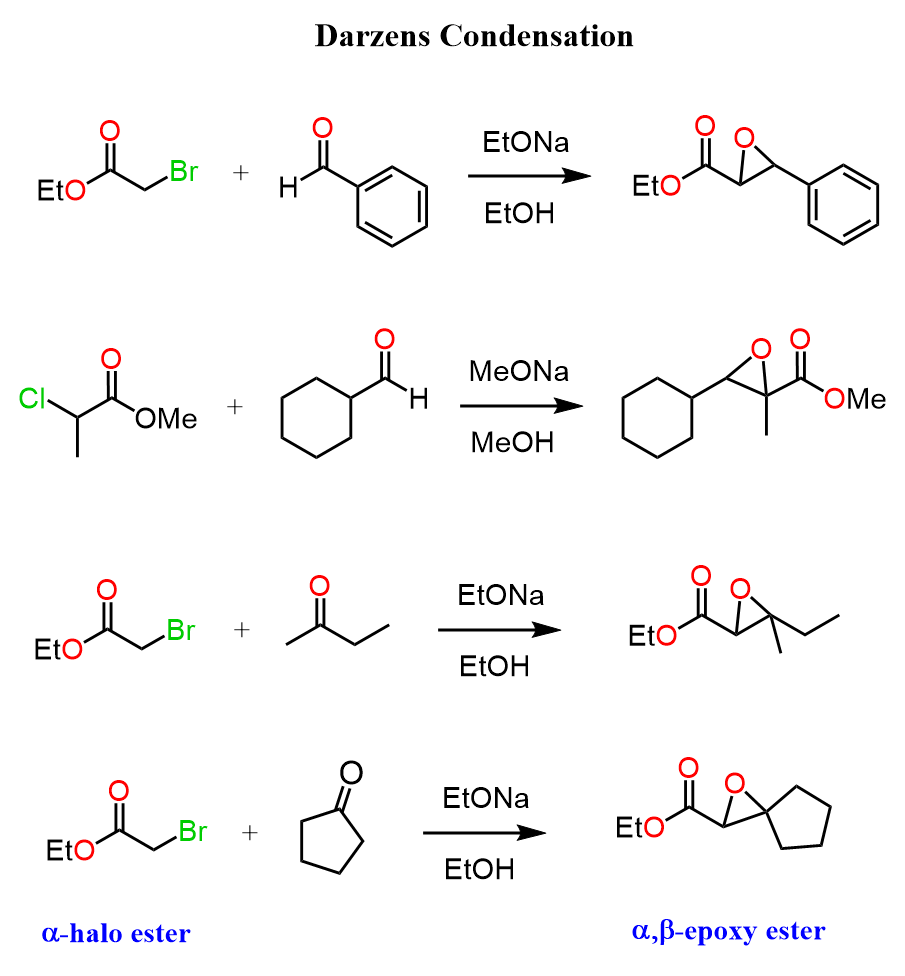
This time, we have a Darzens condensation where the intermediate β-alkoxy carbonyl has a halogen on the α position, which is next to the negatively charged oxygen, and as you may remember, these are common motifs for undergoing intramolecular SN2 reactions to form epoxides.
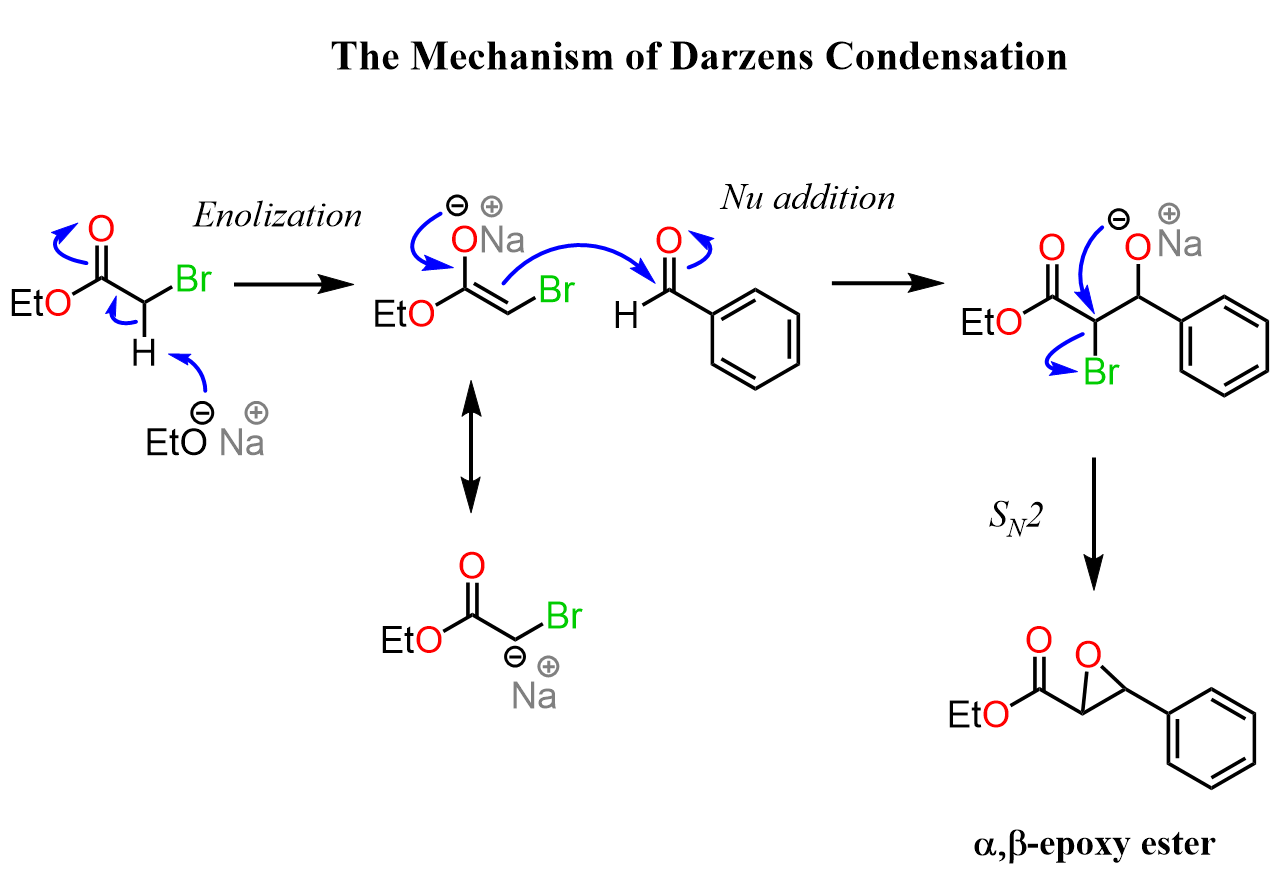
Let’s also put together a complete mechanism for the Darzens condensation:
Alpha halo esters are great tools in organic synthesis, and one such example is the Reformatsky reaction, where they react with aldehydes or ketones in the presence of zinc to form a nucleophilic organozinc intermediate that adds to the carbonyl, ultimately giving β-hydroxy esters after protonation.
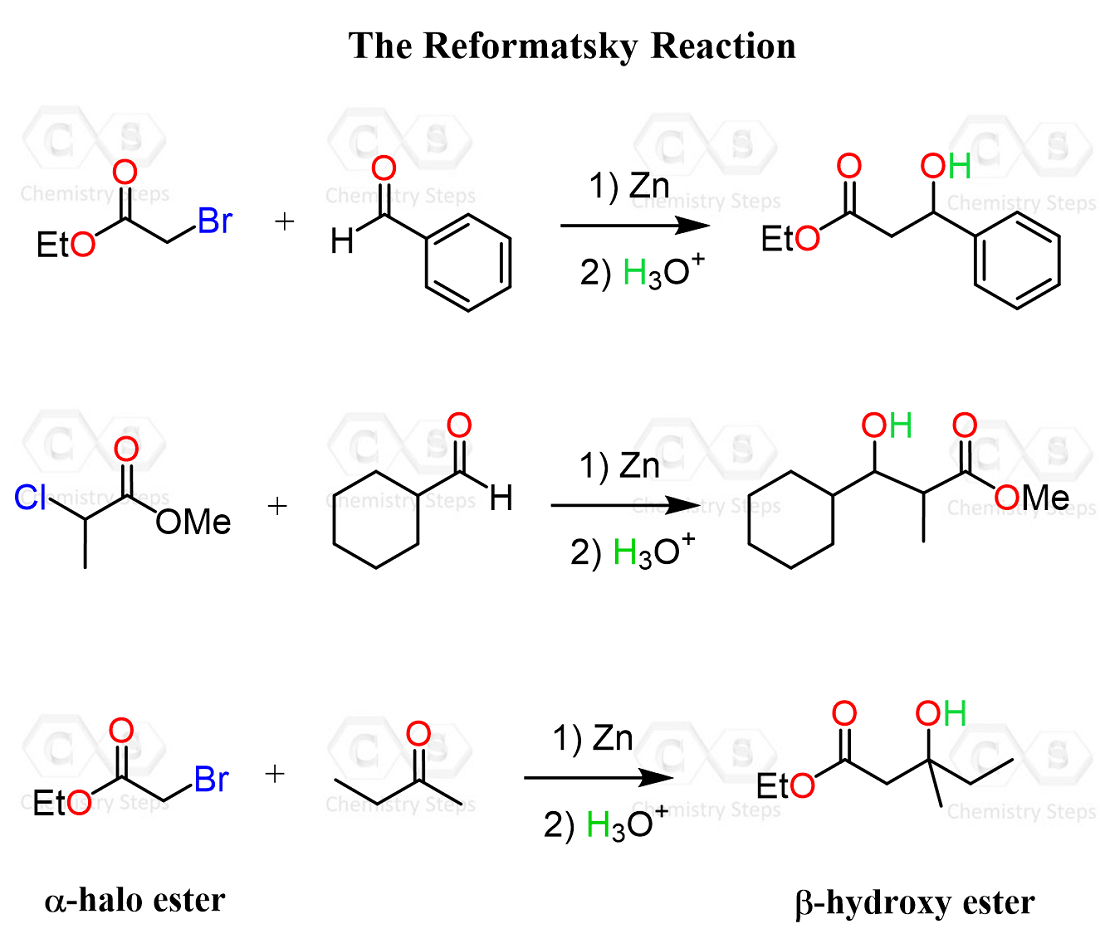
Check out this post for more details and practice problems on the Reformatsky reaction.
Check Also
- Keto-Enol Tautomerization
- Alpha Halogenation of Enols and Enolates
- The Haloform and Iodoform Reactions
- Alpha Halogenation of Carboxylic Acids
- Alpha Halogenation of Enols and Enolates Practice Problems
- The E1cB Elimination Mechanism
- Aldol Reaction – Principles and Mechanism
- Aldol Condensation – Dehydration of Aldol Addition Product
- Intramolecular Aldol Reactions
- Aldol Addition and Condensation Reactions – Practice Problems
- Crossed Aldol And Directed Aldol Reactions
- Crossed Aldol Condensation Practice Problems
- The Cannizzaro reaction
- Alkylation of Enolates Alpha Position
- Enolate Alkylation Practice Problems
- Acetoacetic Ester Synthesis
- Acetoacetic Ester Enolates Practice Problems
- Malonic Ester Synthesis
- Decarboxylation
- Michael Reaction: The Conjugate Addition of Enolates
- Robinson Annulation, Shortcut, and Retrosynthesis
- Claisen Condensation
- Dieckmann Condensation – An Intramolecular Claisen Reaction
- Crossed Claisen and Claisen Variation Reactions
- Claisen Condensation Practice Problems
- Stork Enamine Synthesis
- The Reformatsky Reaction
- Mannich Reaction
- The Knoevenagel Condensation
- Enolates in Organic Synthesis – a Comprehensive Practice Problem
