In Knoevenagel condensation, we take a molecule where a methylene (CH2) is connected to (preferably) two electron-withdrawing groups, so we can deprotonate it and react with an aldehyde or a ketone. This allows the formation of a carbon-carbon double bond between the carbons of the CH2 and the carbonyl groups:
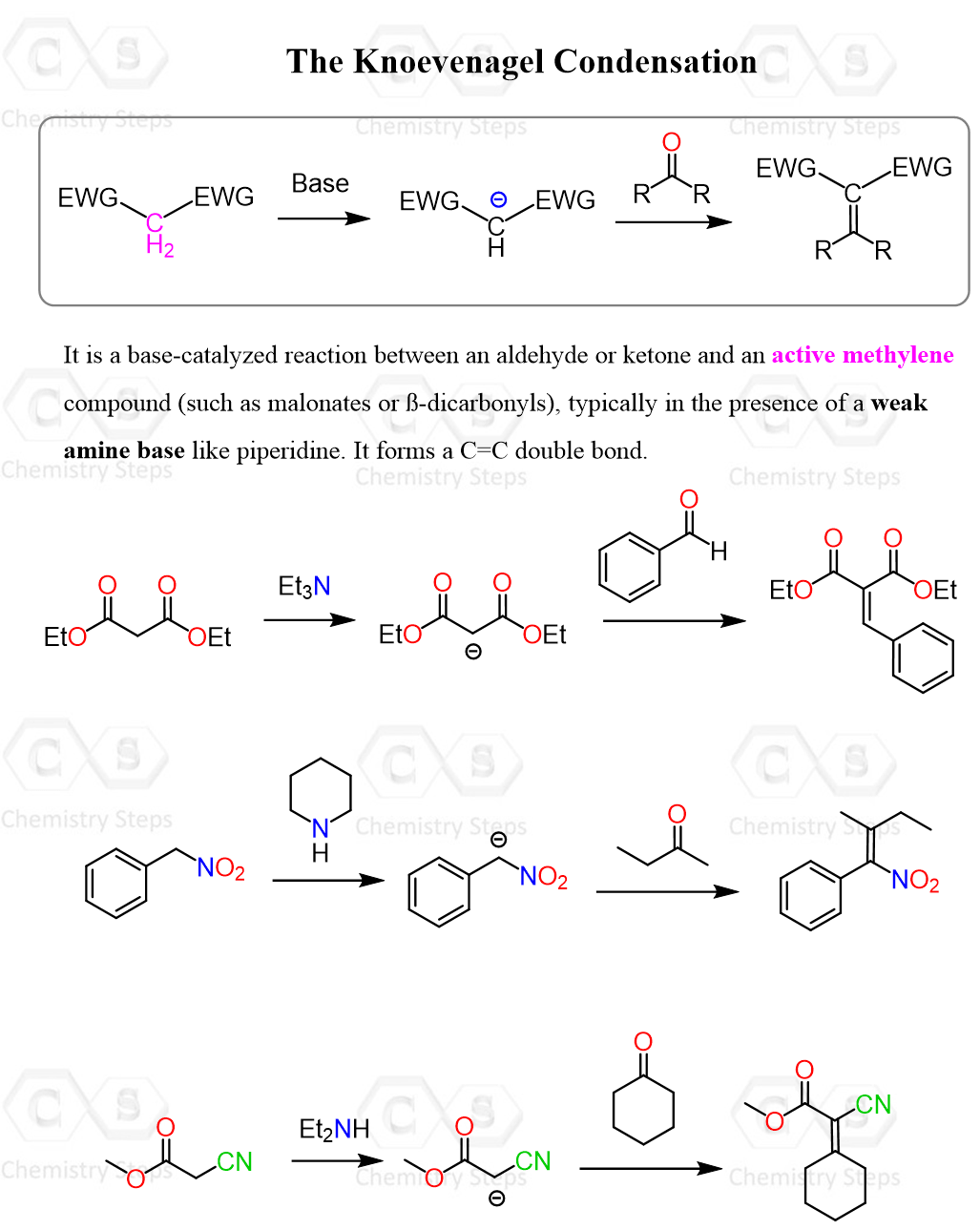
So, it is not very different from the many condensation reactions of enolates, such as the aldol, Claisen, etc. They all follow the same idea of preparing an enolate and reacting with a carbonyl compound. However, there are, of course, specifics to each, so let’s dive a little deeper into the details of the Knoevenagel condensation. The official description can be phrased as: the Knoevenagel condensation occurs between aldehydes or ketones and active methylene compounds in the presence of a weak base such as primary, secondary, or tertiary amines.
Now, one difference we see compared to the aldol reaction, for example, is that a weak base is used instead of stronger ones such as LDA, EtONa, NaH, etc.
The reason for this is exactly the prevention of competing enolate condensation reactions.
The Mechanism of the Knoevenagel Condensation
Let’s consider the first reaction between diethyl malonate and benzaldehyde shown above. Diethyl malonate is a classic dicarbonyl compound with quite acidic protons on the alpha (middle) carbon, so when we treat it with a base such as triethylamine, the conjugate base enolate is formed, which then attacks the carbonyl carbon of the aldehyde. This forms a tetrahedral intermediate, which, upon a proton transfer, forms the alcohol intermediate shown below.
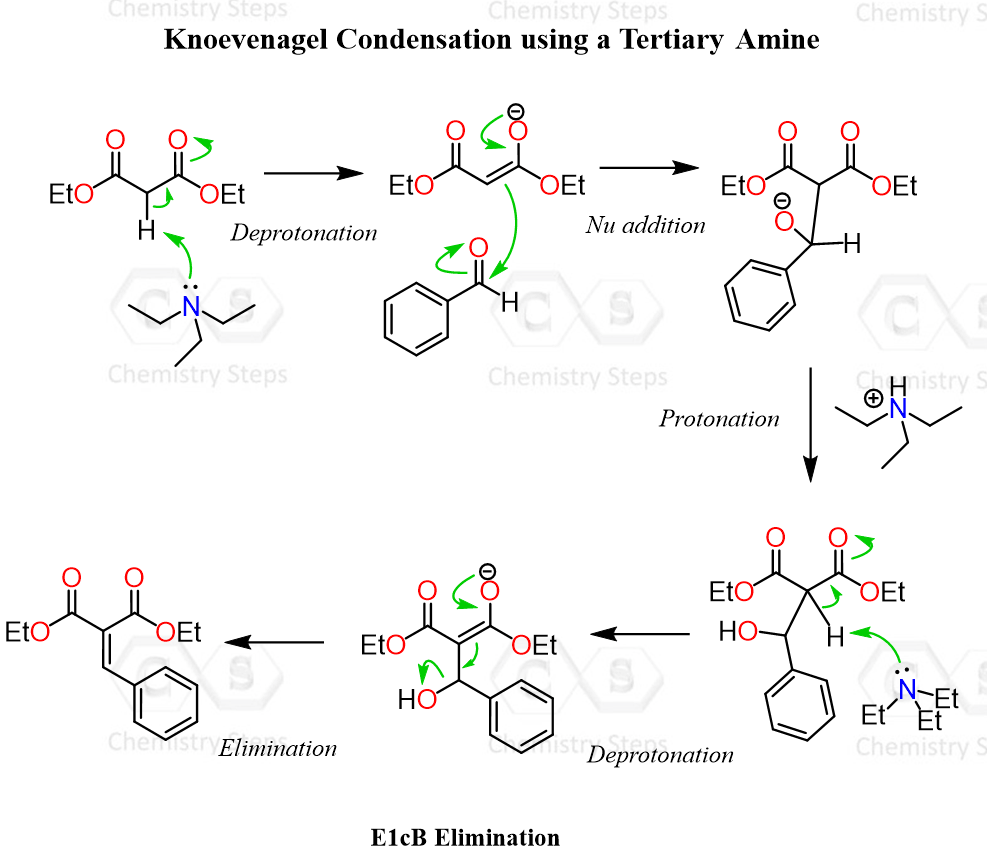
The alcohol then undergoes an E1cB elimination when the base attacks the acidic hydrogen, forming the conjugate base. Remember, in the E1cB elimination, the base first removes a proton to generate a stabilized anion, and only then does the leaving group depart. This stepwise pathway is favored because the intermediate is stabilized by the adjacent electron-withdrawing groups, ultimately leading to the formation of the α,β-unsaturated product.
So, the final product of the reaction is an α,β-unsaturated carbonyl compound.
The Knoevenagel Condensation with Primary and Secondary Amine Catalysis
There is an accepted alternative mechanism for the Knoevenagel condensation when a primary or a secondary amine is used. The difference between these and tertiary or other bulky amines is that they are more nucleophilic, thus capable of attacking the carbonyl group of the aldehyde or the ketone.
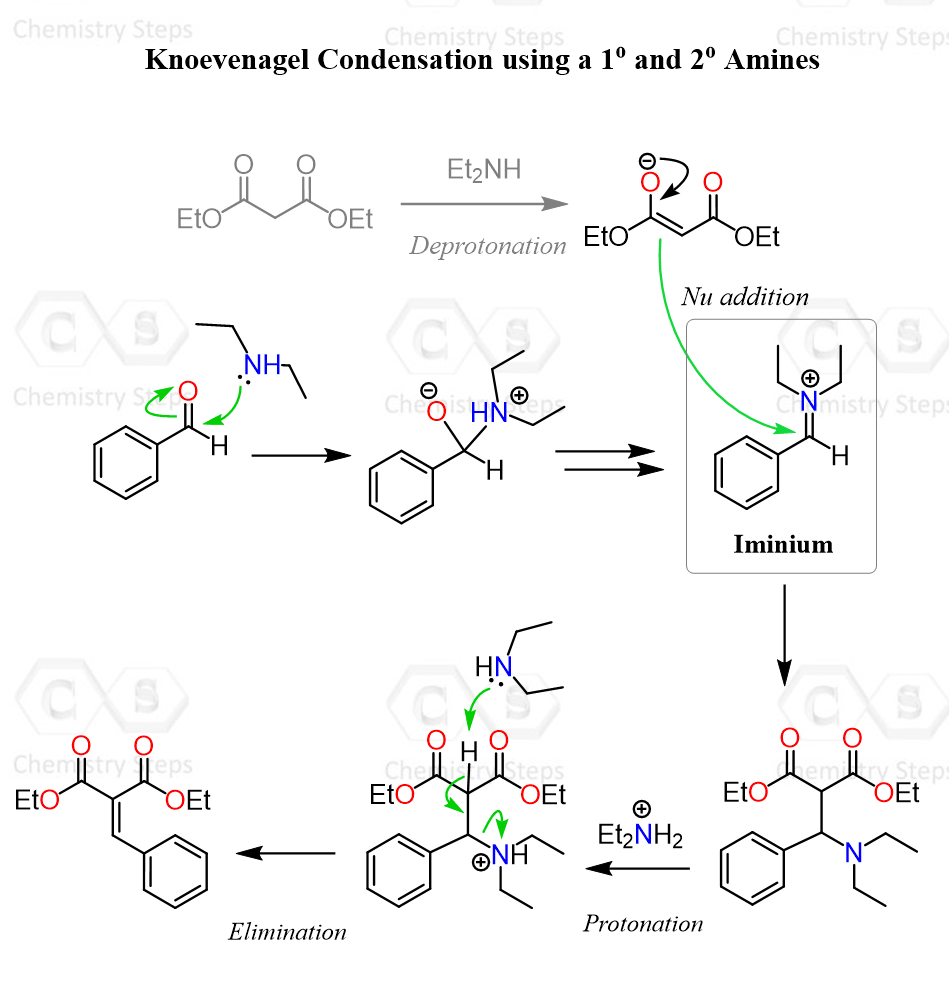
Remember, when aldehydes and ketones react with amines, an iminium ion intermediate is formed, which is very electrophilic. Because of this electrophilicity, the iminium ion is attacked by the conjugate base of diethyl malonate or another active methylene-containing compound, forming the amine-containing intermediate. Remember, this is a Michael addition when the conjugate base of the carbonyl performs a 1,4-addition.
The nitrogen is then protonated, which allows the base to perform an E2 elimination and give the desired Knoevenagel product.
Summary
The Knoevenagel condensation is a versatile carbon-carbon bond-forming reaction that relies on the reactivity of active methylene compounds and mild base conditions. While it follows the same general idea as other enolate condensations such as the aldol reaction, its use of weak amine bases helps avoid competing side reactions and provides better control.
Mechanistically, the reaction can proceed through direct enolate addition to a carbonyl compound followed by E1cB elimination, or through an alternative pathway involving iminium ion formation when primary or secondary amines are used. In both cases, the key outcome is the formation of an alpha-beta unsaturated carbonyl compound.
Overall, the Knoevenagel condensation is an important and widely used transformation in organic synthesis, offering a reliable method for constructing conjugated systems from simple starting materials.
Check Also
- Keto-Enol Tautomerization
- Alpha Halogenation of Enols and Enolates
- The Haloform and Iodoform Reactions
- Alpha Halogenation of Carboxylic Acids
- Alpha Halogenation of Enols and Enolates Practice Problems
- The E1cB Elimination Mechanism
- Aldol Reaction – Principles and Mechanism
- Aldol Condensation – Dehydration of Aldol Addition Product
- Intramolecular Aldol Reactions
- Aldol Addition and Condensation Reactions – Practice Problems
- Crossed Aldol And Directed Aldol Reactions
- Crossed Aldol Condensation Practice Problems
- The Cannizzaro reaction
- Alkylation of Enolates Alpha Position
- Enolate Alkylation Practice Problems
- Acetoacetic Ester Synthesis
- Acetoacetic Ester Enolates Practice Problems
- Malonic Ester Synthesis
- Decarboxylation
- Michael Reaction: The Conjugate Addition of Enolates
- Robinson Annulation, Shortcut, and Retrosynthesis
- Claisen Condensation
- Dieckmann Condensation – An Intramolecular Claisen Reaction
- Crossed Claisen and Claisen Variation Reactions
- Claisen Condensation Practice Problems
- Stork Enamine Synthesis
- The Reformatsky Reaction
- Mannich Reaction
- Enolates in Organic Synthesis – a Comprehensive Practice Problem
