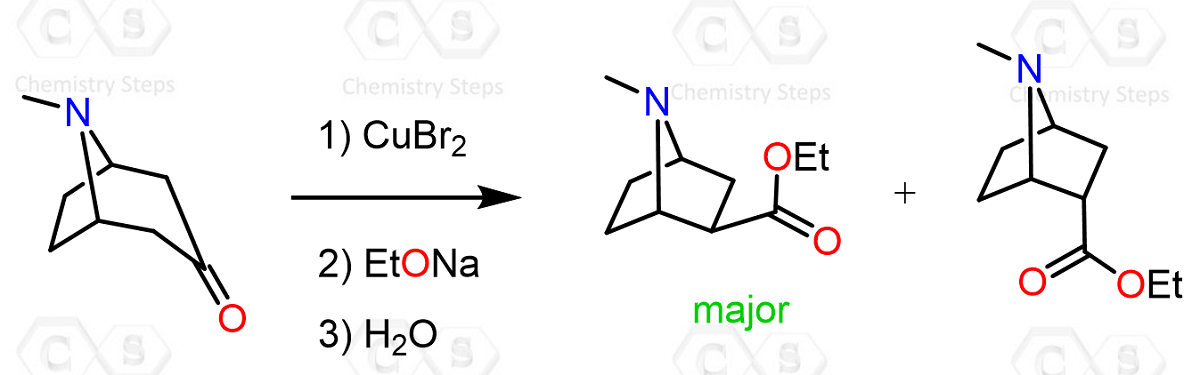
The Favorskii rearrangement is a base-promoted skeletal rearrangement of α-halo ketones possessing at least one α-hydrogen. Treatment with base in the presence of a nucleophile (such as alcohols, amines, or water) results in a rearrangement via a cyclopropanone intermediate, ultimately giving carboxylic acids or carboxylic acid derivatives (esters or amides). It is widely used for the synthesis of highly branched carboxylic acids.
Let’s consider the ring contraction of 2-bromocyclohexanine:
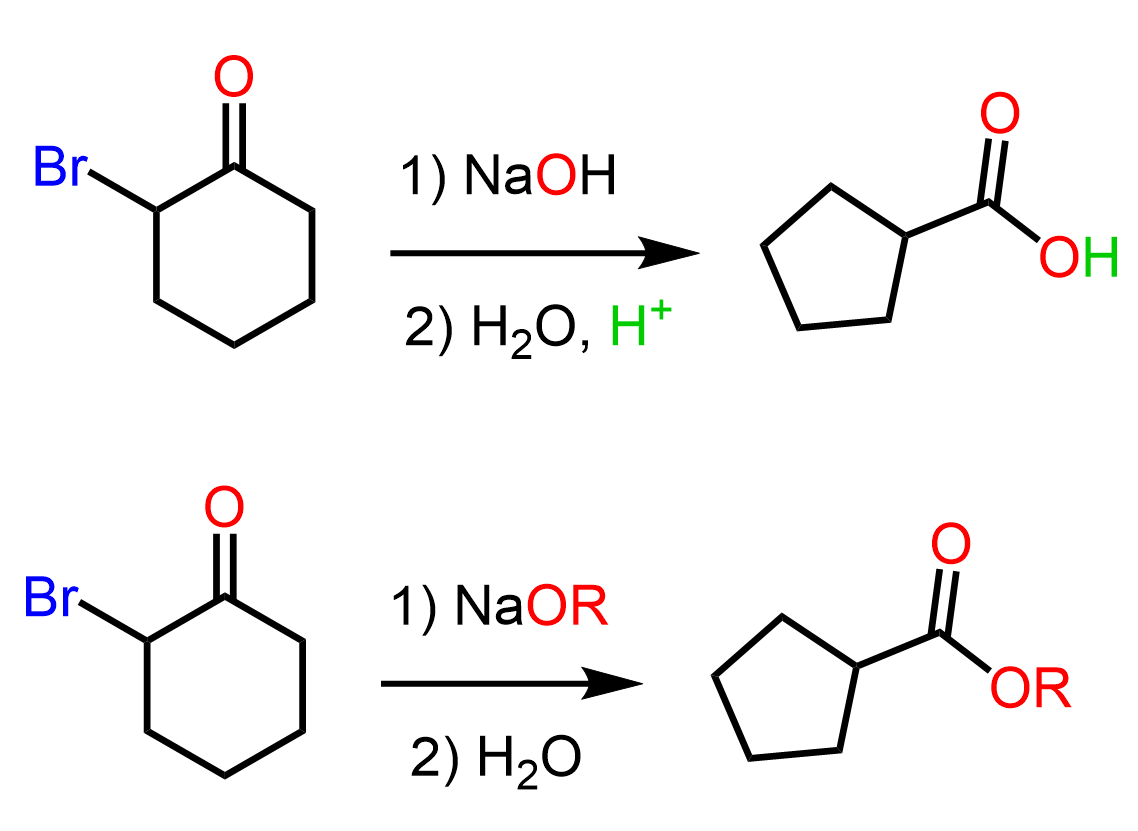
The halogen can be chlorine, bromine, or iodine, while the base is usually an alkoxide or hydroxide. Upon rearrangement, acyclic α-halo ketones give acyclic carboxylic acid derivatives, while cyclic α-halo ketones undergo a ring-contraction reaction to afford a cyclic carboxylic acid derivative that is one carbon smaller.
We know that the α position of the carbonyl group is significantly more acidic (pKa ~ 19) than other positions, so when treated with sodium hydroxide or an alkoxide, a deprotonation occurs, forming the corresponding enolate ion. (Yes, if you are wondering how a hydroxide can deprotonate a ketone, given that the pKa of water is ~16, the answer is that the enolate is formed in very small amounts, and the reaction proceeds as the enolate is continuously consumed in the next step. This drives the equilibrium forward.)
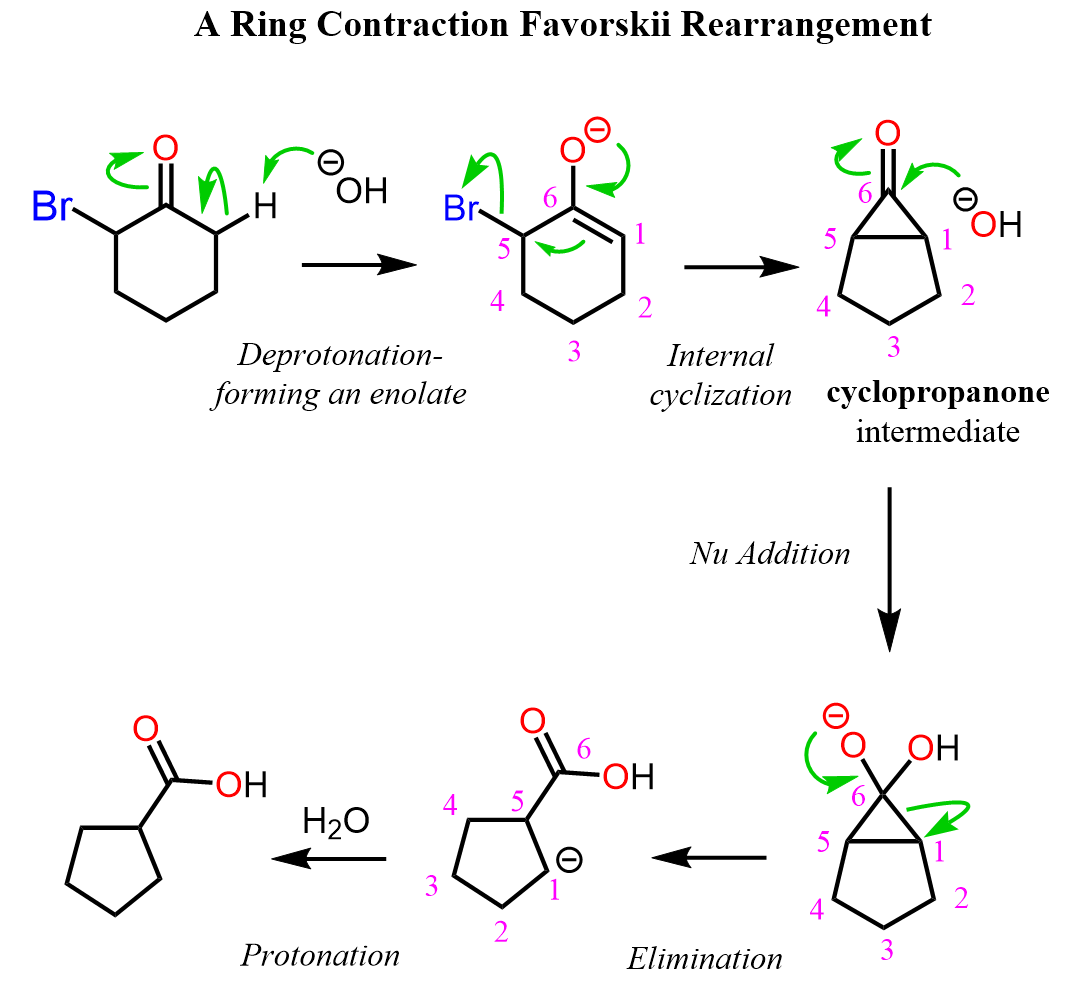
The enolate then undergoes an internal cyclization, forming a highly strained cyclopropanone intermediate. This intermediate is opened by nucleophilic attack of hydroxide or alkoxide, followed by an addition-elimination sequence.
In the final step, the carboxylate is protonated to give the corresponding carboxylic acid or ester, and in cyclic systems, a new one-carbon smaller ring is formed.