Parent chain is the “main part” of the molecule. Just like a car or a house is the main component, no matter what accessories and decorations we can have on them.
For example, suppose we have an infinitely long carbon chain, ok, maybe not infinitely, but let’s say 30 carbons:

If we add smaller fragments (substituents) to it, it will still be the main component of the molecule:

This is evident without even going into the rules of identifying the parent chain. Simply put, the longest chain is the parent chain. So, let’s add some common alkyl groups to get familiar with their nomenclature:

Now, of course, we know that things will get more complex in real life (on the test), and you will need to put some more effort into finding the parent chain and naming the molecule.
So, where do things get more complicated?
Let’s say we have decane, and we add a pentyl substituent somewhere in the middle of the chain:

What is the parent chain of this new molecule? This does not look as obvious as the previous example, as there is some visual symmetry, and we need to determine whether it makes any difference if we count the longest carbon chain from the left or from the right.
Oh, by the way, the official definition of the parent chain is the longest continuous carbon chain that is present in the molecule. At least for now, we are good to go with this.
Take a moment and try to find the parent chain by numbering from both ends of the molecule.
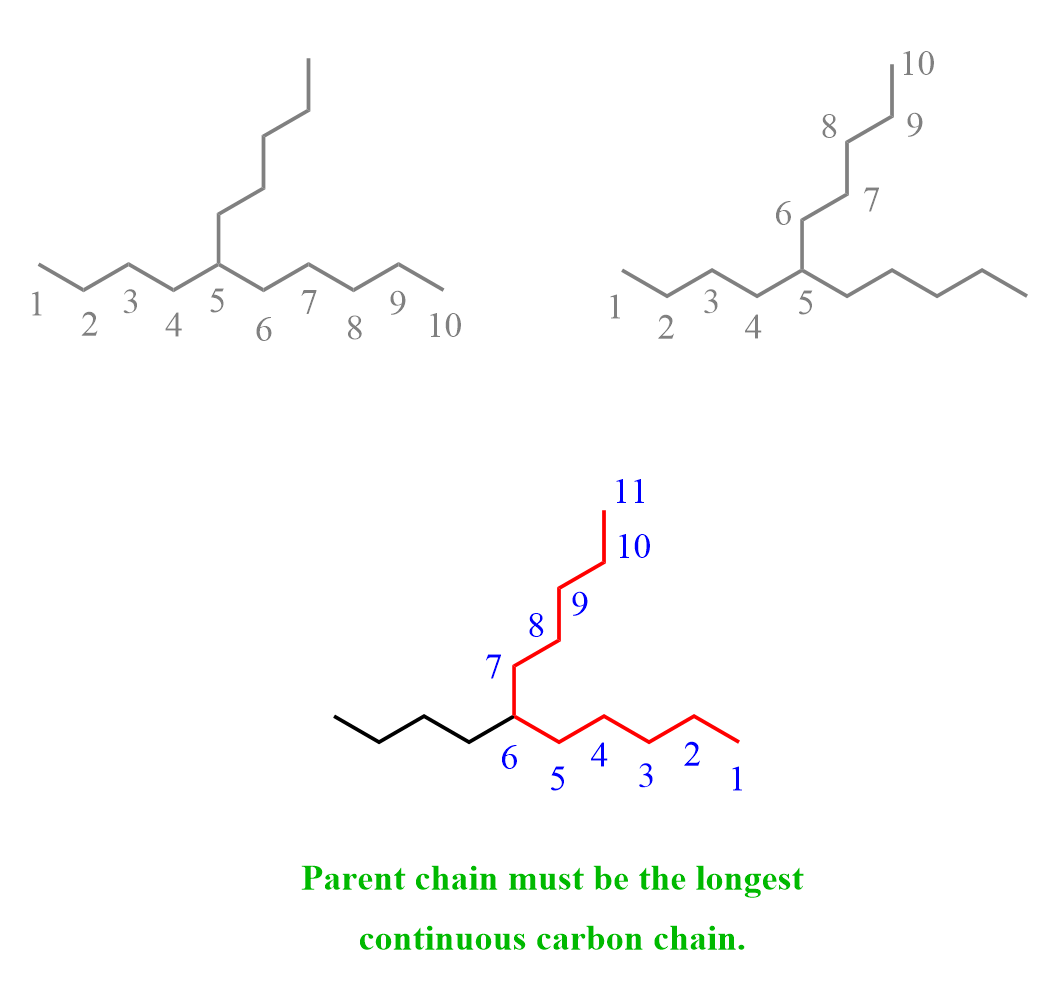
Notice that in the highlighted version, the parent chain has 11 carbons (undecane) while in the other two, it is only 10 carbons (10).
The alkyl group that is not part of the parent chain is n-butyl or simply butyl substituent, which we add to the beginning of the name by mentioning its position with a number and a hyphen:

We have a separate post dedicated to the IUPAC nomenclature of alkanes and haloalkanes, so check that out for more examples and details on naming rules in organic chemistry, but for now, let’s consider a few more scenarios where finding the parent chain is not so trivial.
The Number of Substituents on the Parent Chain
Sometimes, there might be more than one way of having a parent chain, as the longest carbon chain is the same regardless of how we number it. For example, the parent chain in the following molecule is nonane no matter how we number it. In these types of situations, you need to choose the one with the greater number of substituents:

Notice that in the correct option, there are 3 substituents, while the other two have only 2.
Parent Chain in Cycloalkanes
There might be some conflict of information on how we determine if the ring is the parent chain or the longer carbon chain in the molecule. In most textbooks, the priority is given to whatever has the greatest number of carbons, and that’s how we do it at Chemistry Steps, too. Once again, we are talking about a situation where the number of carbons in the ring and the open chain – whichever is greater gets to be the parent chain:

Notice that the carbons in the ring belong to the ring only. I.e., you cannot count the carbon twice or include it in the carbon chain.
Now, about the conflict of information. In a relatively recent update of IUPAC rules (IUPAC blue book 2013), it is stated that the ring has a priority o mater the length of the open chain.
Quoting the IUPAC 2013 Blue Book:
P-44.1.2.2 – Ring Systems composed of rings and chains (exclusive of linear phanes)
Two methods are recognized to name systems composed of rings and chains (exclusive of linear phanes).
(1) Within the same class, a ring or ring system has seniority over a chain. When a ring and a chain contain the same senior element, the ring is chosen as the parent. Rings and chains are chosen regardless of their degree of hydrogenation. As a consequence, this approach prefers the choice of a ring over a chain in systems composed of cyclic and acyclic hydrocarbons. For example, they have heptylbenzene as a preferred IUPAC name even though the chain has more carbon atoms. One nuance here is that benzene is an aromatic compound, so one may think that’s what gives it a priority; however, it is mentioned in the rule that “Rings and chains are chosen regardless of their degree of hydrogenation.”
So, apparently, hexylcyclopentane is the preferred IUPAC name (PIN) for what we called cyclopentylhexane.
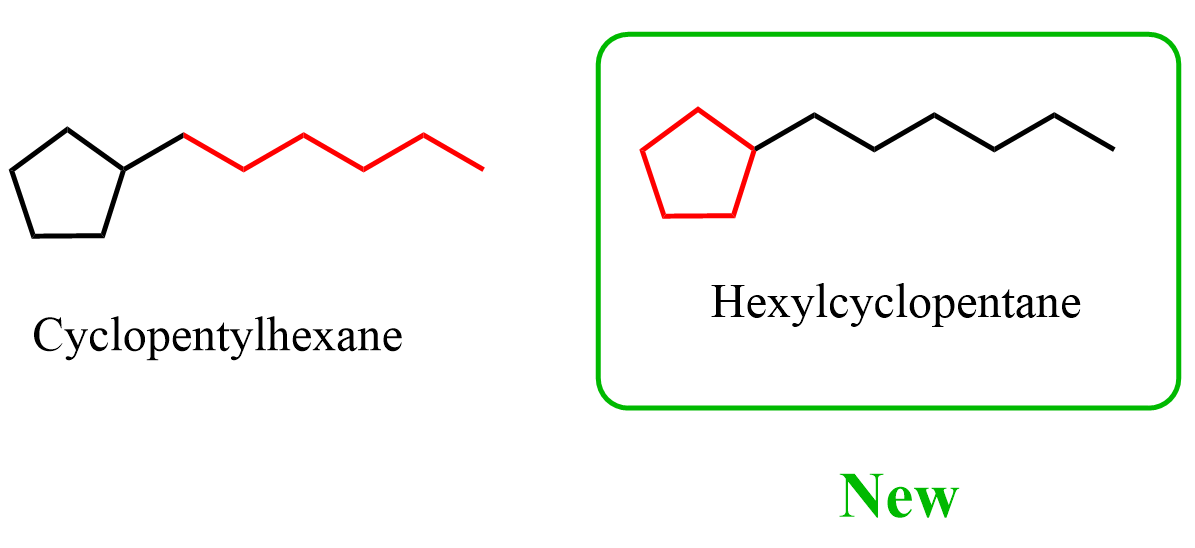
So, which rules do we follow when naming certain alkanes, cycloalkanes, and alkenes? Honestly, I don’t think this is the most critical part of learning and understanding organic chemistry… In most examples and practice problems on Chemistry Steps, we stick with the earlier rules, since that’s what most textbooks, at least in the US, tend to use. At the end of the day, it’s up to your instructor how they want to handle these cases, so be sure to follow the guidelines from your class or check with them if anything’s unclear.
This is all I wanted to cover on what the parent chain is and how to find it. The overall name of the compound often depends on the position and numbering of the substituents, too, so check this comprehensive article on IUPAC nomenclature for a more detailed discussion.



