The SN2 (Substitution Nucleophilic Bimolecular) reaction involves a single concerted step, where the nucleophile attacks the carbon bearing the leaving group from the backside, and the leaving group departs simultaneously.
As an example, let’s show the reaction between 2-bromobutane and the cyanide ion:
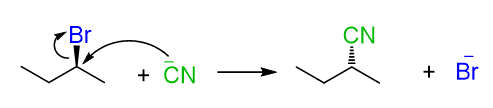
For this substitution to occur, the nucleophile must access the carbon connected to the leaving group bromide. Therefore, the idea behind making alkyl halides more reactive in SN2 substitution boils down to two main factors:
- Make the carbon bearing the leaving group more accessible
- Make the carbon–halogen bond break more easily
Let’s start with the accessibility of the carbon atom, which is its steric hindrance.
Steric Hindrance and SN2 Reactivity
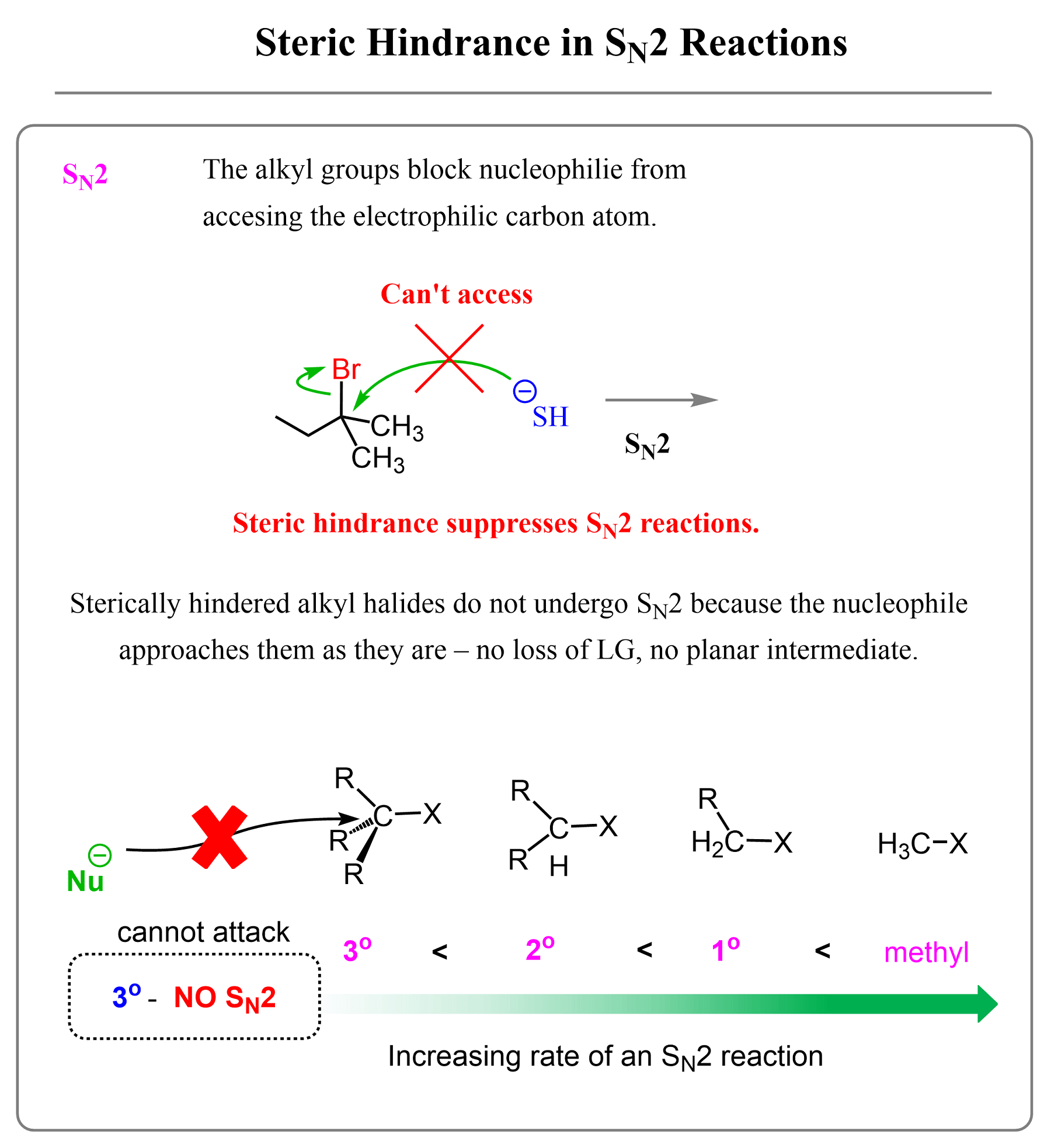
The most reactive alkyl halides, or other substrates such as mesylates and tosylates, are the methyl substrates. The carbon in the methyl group is connected to three hydrogens, and there are no carbon atoms blocking the access for the nucleophile. After this, we have the ethyl substrates, and the reactivity keeps decreasing with any additional alkyl groups.
Here is a table illustrating the SN2 reaction rate of methyl and other alkyl bromides towards iodide ion.

You will likely not need to remember the numbers, but understanding the reactivity of alkyl halides in substitution reactions will be very helpful at some point when we start talking about determining whether the reaction is SN2, SN1, or perhaps E1 and E2 eliminations need to be considered as well.
For a better visualization, remember that tertiary alkyl halides never undergo SN2 substitution because of the three alkyl groups blocking the electrophilic carbon:
On a side note, remember that methyl and primary alkyl halides never participate in SN1 reactions as the intermediate carbocation is too unstable to form.
Combining these trends, you need to keep in mind the extreme cases of alkyl halides in SN1 and SN2 reactions:
- Tertiary alkyl halides never react by the SN2 mechanism.
- Primary alkyl halides never react by the SN1 mechanism.

Because the reaction depends on both the nucleophile and the alkyl halide, the structure of the alkyl halide plays a crucial role in determining its reactivity.
The Effect of the Leaving Group in SN2 Reactions
The other factor, aside from the steric hindrance, affecting the reactivity of alkyl halides is how easily the C-halogen bond is broken. In other words, we are comparing the leaving ability of the halide-the easier the C–X bond cleavage, the better the leaving group, or we can also say the better the leaving group, the faster the SN2 reaction.

Among the halides, iodide is the best leaving group, followed by bromine and chlorine: I⁻ > Br⁻ > Cl⁻ > F⁻
The most common leaving groups in nucleophilic substitution and elimination reactions are shown below:

Notice that the stability of the leaving group has to do with the pKa value of the corresponding acid. The stronger the acid, the more stable (weaker) its conjugate base is, thus the better leaving group it is.
The Effect of Solvent in SN2 Reactions
Although this is not directly related to the alkyl halide, you also need to remember that polar aprotic solvents, such as acetone, dimethyl sulfoxide (DMSO), dimethylformamide (DMF), and acetonitrile, are best suited for SN2 reactions.
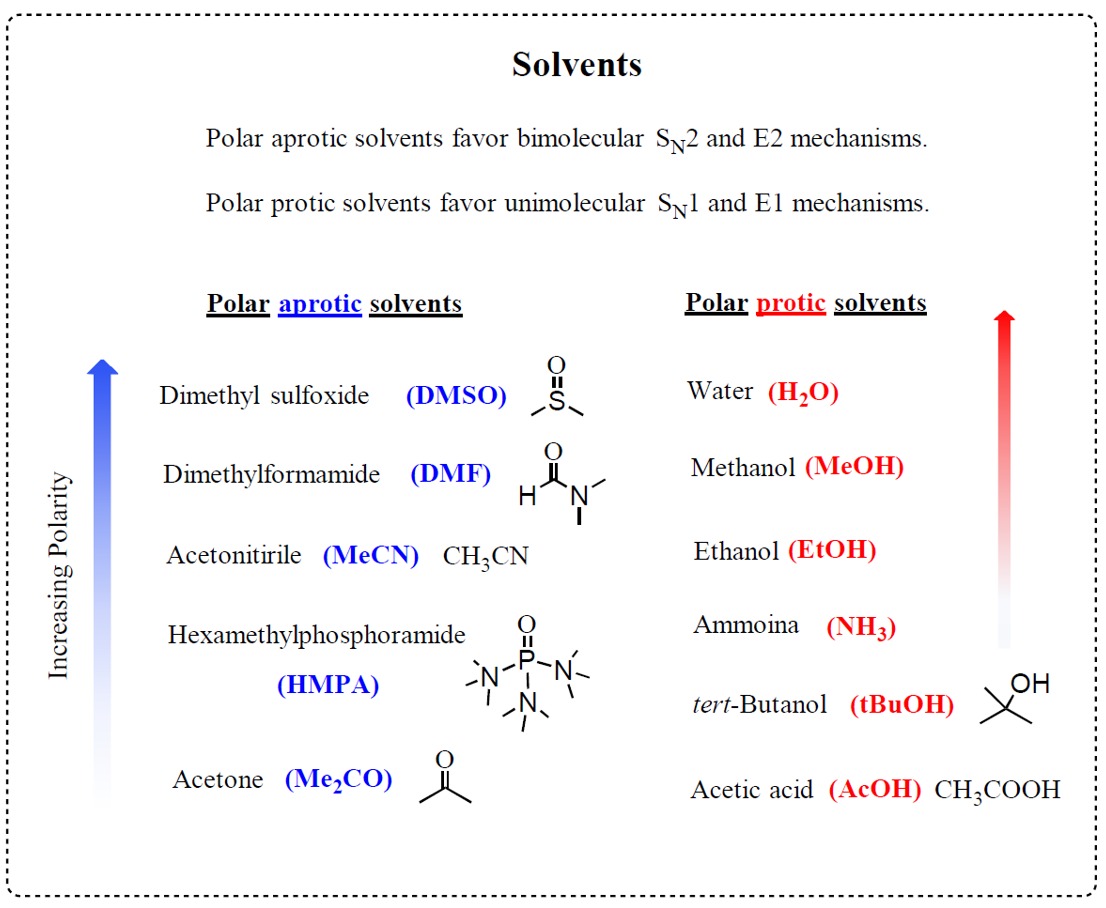
These solvents are polar enough to dissolve ionic species like the nucleophile and leaving group, but they do not form strong hydrogen bonds with the nucleophile. As a result, the nucleophile remains “free” and highly reactive, able to attack the electrophilic carbon more readily.
For example, if we use NaCl as a nucleophile for an SN2 reaction in DMSO, the solvent molecules surround only the counterion Na+, whereas the chloride ion is free to attack the electrophilic carbon atom:

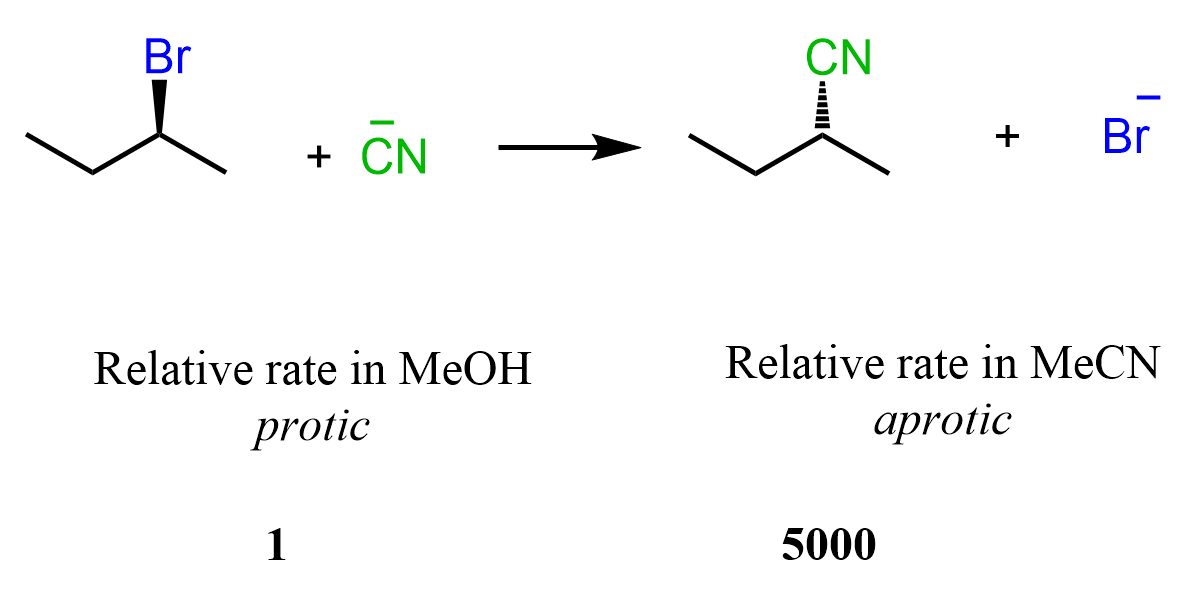
This principle applies to any nucleophile, and we can see how the aforementioned reaction between 2-bromobutane and cyanide ion proceeds 500 times faster in DMSO than in methanol, which is a polar protic solvent.
In contrast, polar protic solvents (like water, alcohols, and carboxylic acids) tend to solvate nucleophiles through hydrogen bonding, effectively surrounding and stabilizing them. This reduces their nucleophilicity and slows or even prevents the SN2 reaction.
Check Also
- Introduction to Alkyl Halides
- Nomenclature of Alkyl Halides
- Substitution and Elimination Reactions
- Nucleophilic Substitution Reactions – An Introduction
- All You Need to Know About the SN2 Reaction Mechanism
- SN2 Mechanism: Kinetics, Thermodynamics, Curved Arrows, and Stereochemistry with Practice Problems
- The Stereochemistry of SN2 Reactions
- Stability of Carbocations
- The SN1 Nucleophilic Substitution Reaction
- Reactions of Alkyl Halides with Water
- The Stereochemistry of the SN1 Reaction Mechanism
- The SN1 Mechanism: Kinetics, Thermodynamics, Curved Arrows, and Stereochemistry with Practice Problems
- Steric Hindrance in SN2 and SN1 Reactions
- Carbocation Rearrangements in SN1 Reactions with Practice Problems
- Ring Expansion Rearrangements
- Ring Contraction Rearrangements
- When Is the Mechanism SN1 or SN2?
- SN1 vs E1 Reactions
- SN2 vs E2 Reactions
- Reactions of Alcohols with HCl, HBr, and HI Acids
- SOCl2 and PBr3 for Conversion of Alcohols to Alkyl Halides
- Alcohols in SN1 and SN2 Reactions
- How to Choose Molecules for Doing SN2 and SN1 Synthesis–Practice Problems
- Exceptions in SN2 and SN1 Reactions
