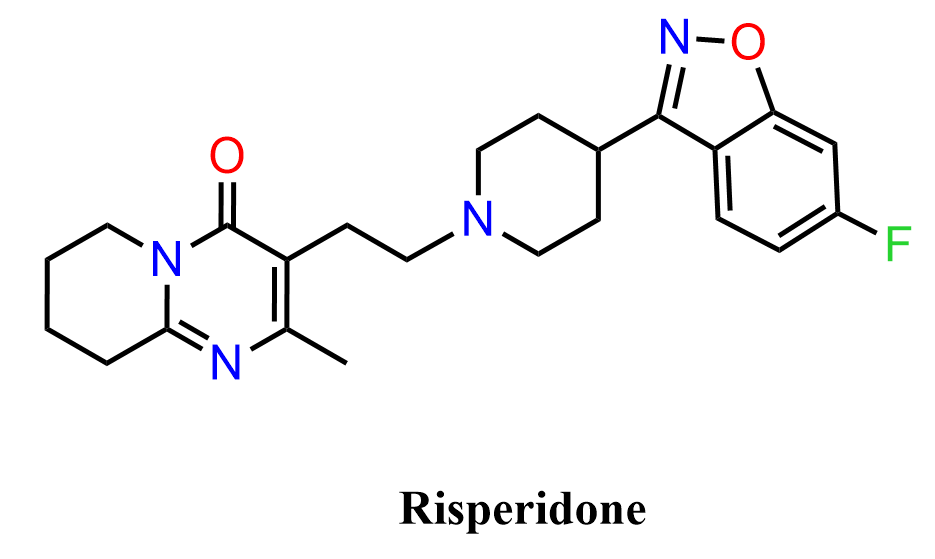
 Risperidone is an atypical antipsychotic medication widely used in the treatment of schizophrenia, bipolar disorder, and irritability associated with autism spectrum disorder. It belongs to a class of drugs known as second-generation antipsychotics, which act primarily by modulating dopamine (D₂) and serotonin (5-HT₂A) receptors in the brain. Structurally, risperidone is a complex molecule containing several important heterocyclic and aromatic fragments, including a benzisoxazole ring, a fluorinated aromatic ring, and a piperidine-containing side chain. Because of this structural complexity, risperidone provides an interesting example of how common organic reactions and functional group transformations can be combined in a multistep synthesis to construct a pharmaceutically relevant molecule.
Risperidone is an atypical antipsychotic medication widely used in the treatment of schizophrenia, bipolar disorder, and irritability associated with autism spectrum disorder. It belongs to a class of drugs known as second-generation antipsychotics, which act primarily by modulating dopamine (D₂) and serotonin (5-HT₂A) receptors in the brain. Structurally, risperidone is a complex molecule containing several important heterocyclic and aromatic fragments, including a benzisoxazole ring, a fluorinated aromatic ring, and a piperidine-containing side chain. Because of this structural complexity, risperidone provides an interesting example of how common organic reactions and functional group transformations can be combined in a multistep synthesis to construct a pharmaceutically relevant molecule.
In this practice problem, we will discuss how to synthesize risperidone starting from benzene.
As with all examples in this section, this discussion is presented solely for educational purposes and is not intended to represent a practical or real-world method of preparation.
Practice
The scheme below shows the brief synthetic transformation, and you can see that there is a fluorobenzene component in the structure.
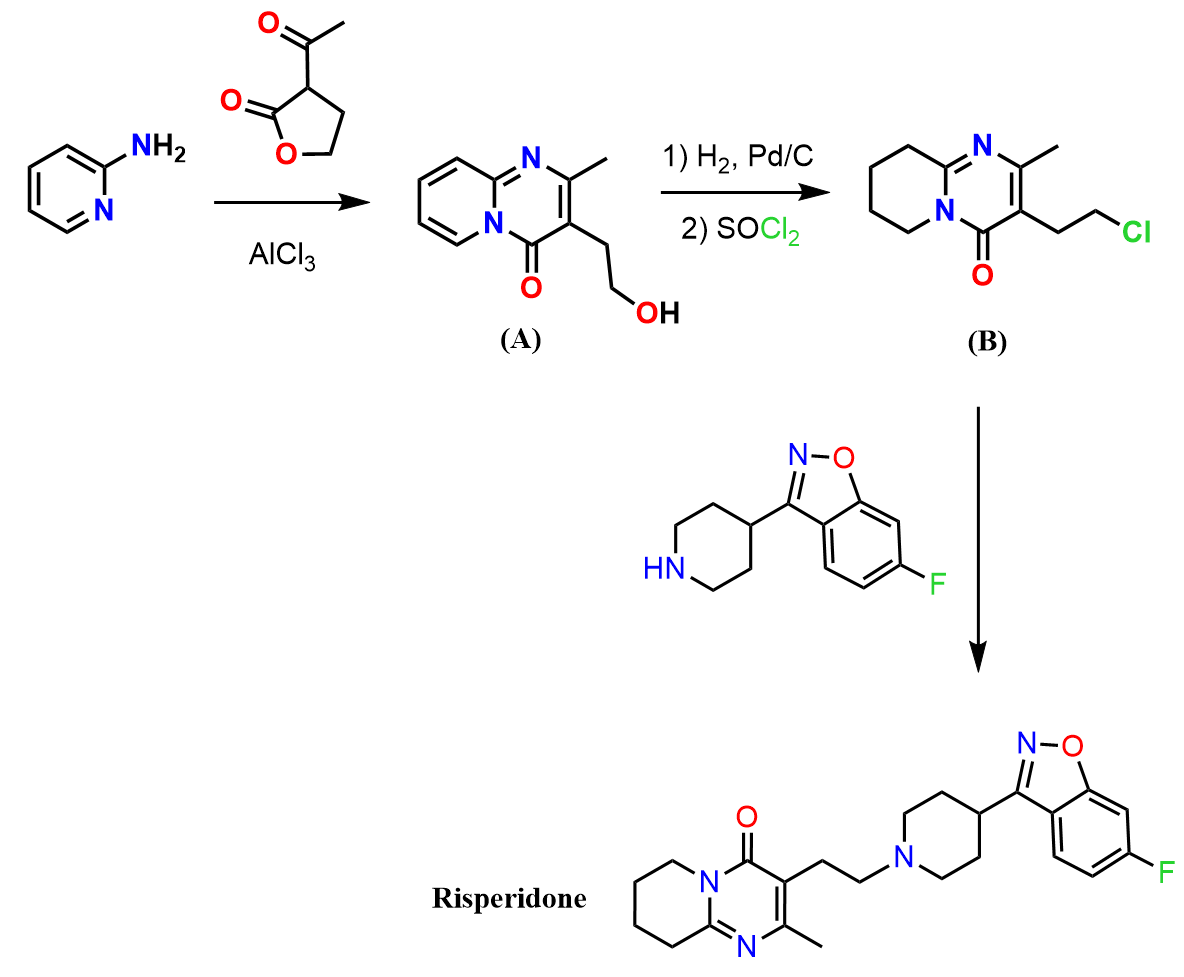
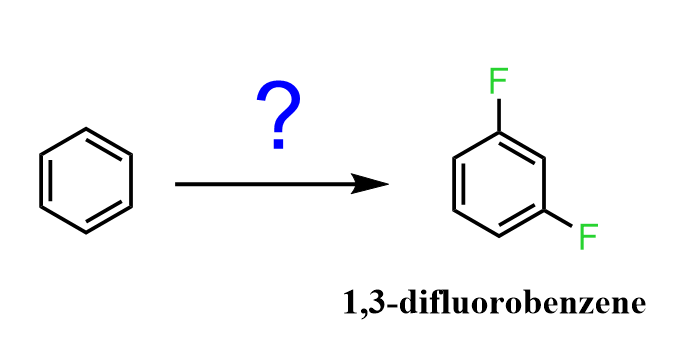
1) Therefore, the first question to think about is how to prepare 1,3-difluorobenzene from benzene.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
