At the beginning of your organic chemistry journey, you’re told a golden rule: carbon likes to have four bonds. This is true almost all the time. Why almost? Because you should never draw a carbon with more than four bonds. That rule is absolute.
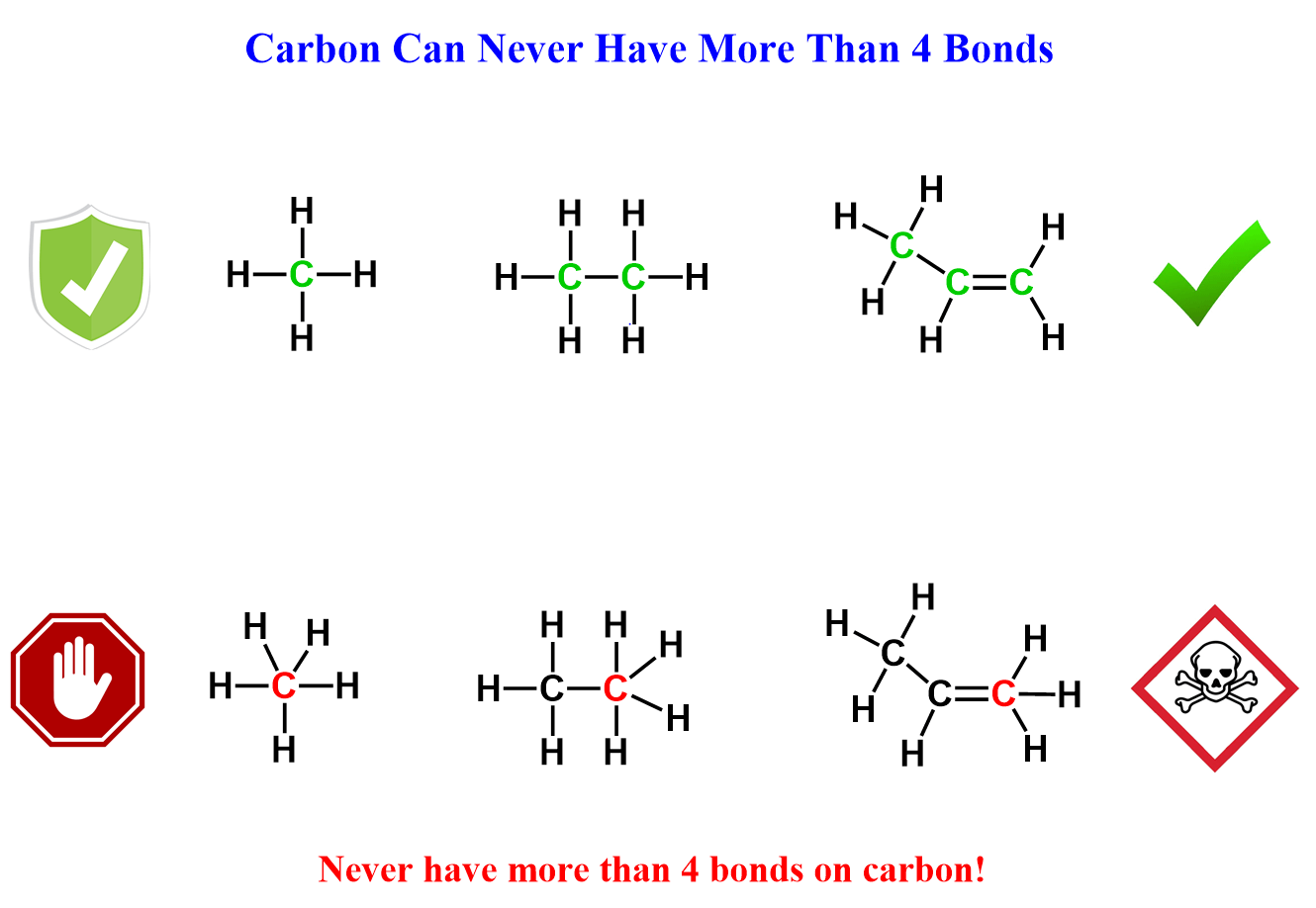
In neutral molecules, carbon always has four bonds, and these can be either four single bonds or a multiple bond such as a double or triple bond. The image below summarizes the common bonding patterns of carbon, nitrogen, oxygen, and halogens in neutral molecules:
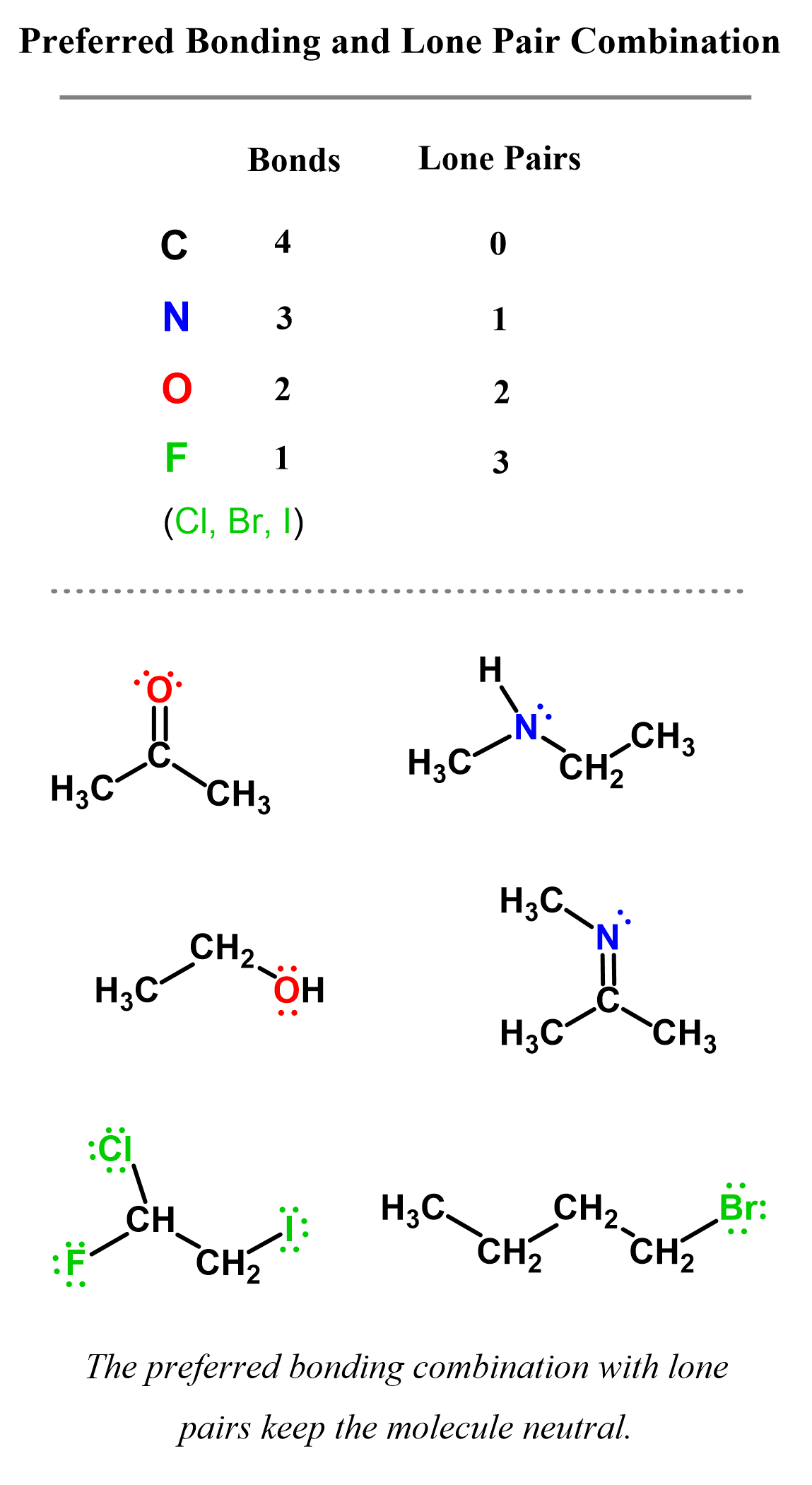
Check the article “Bonding Patterns in Organic Chemistry” for more details and how lone pairs and formal charges come to the rescue when these atoms do not follow their standard bonding pattern.
Now, the question you were searching:
Can carbon have three bonds?
The answer is yes, carbon can have three bonds, but we need to know when and how that happens. In short, this is possible with the right combination of bonds, lone pair, and a formal charge. There are two possibilities for carbon having three bonds, and that is either in carbocations or carbanions.
In carbocations, there is a carbon that is positively charged because it only has three bonds and no lone pair. Carbocations do not satisfy the octet rule, making them very unstable and reactive. They’ll quickly seek electrons from nearby atoms or molecules and react with them to form new molecules.
Carbanions contain a carbon with three bonds and a lone pair which makes them negatively charged. Unlike carbocations, carbanions do satisfy the octet rule, but they are still unstable because they carry excess electron density. They’ll look to react with electron-poor species (electrophiles).
Here are some examples of molecules containing carbon atoms with three bonds. Pay attention to the number of lone pairs and formal charges:
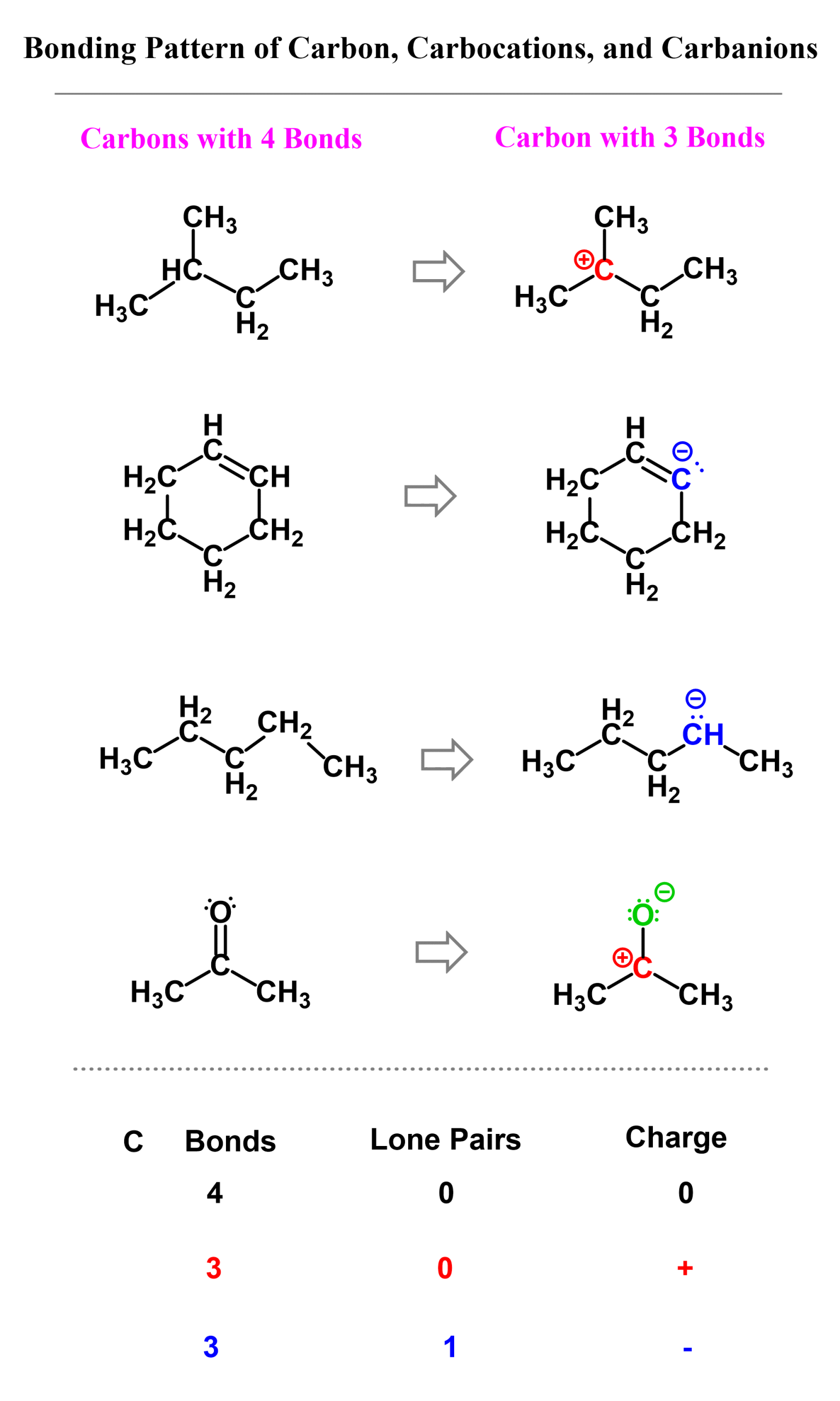
Check the mentioned article for bonding patterns and examples that include second-row elements nitrogen, oxygen, fluorine, and the rest of the halogens.
Check Also
- Lewis Structures in Organic Chemistry
- Valency and Formal Charges in Organic Chemistry
- Bonding Patterns in Organic Chemistry
- How to Determine the Number of Lone Pairs
- sp3, sp2, and sp Hybridization in Organic Chemistry with Practice Problems
- How to Quickly Determine The sp3, sp2, and sp Hybridization
- Bond Lengths and Bond Strengths
- VSEPR Theory – Molecular and Electron Geometry of Organic Molecules
- Dipole-dipole, London Dispersion, and Hydrogen Bonding Interactions
- Dipole Moment and Molecular Polarity
- Boiling Point and Melting Point in Organic Chemistry
- Boiling Point and Melting Point Practice Problems
- Solubility of Organic Compounds
- General Chemistry Overview Quiz
- Bond-Line or Skeletal Structures
- Functional Groups in Organic Chemistry with Practice Problems
- Bond-line, Lewis and Condensed Structures with Practice Problems
- Curved Arrows with Practice Problems
- Resonance Structures in Organic Chemistry with Practice Problems
- Rules for Drawing Resonance Structures
- Bonding Patterns in Organic Chemistry
- Major and Minor Resonance Contributor
- Significant Resonance Structures
- How to Choose the More Stable Resonance Structure
- Drawing Complex Patterns in Resonance Structures
- Localized and Delocalized Lone Pairs with Practice Problems
- Molecular Representations Quiz
