Carbon is positively charged when, instead of the preferred four bonds, it has three bonds. Remember, from the bonding patterns in organic chemistry, carbon prefers to have four bonds and no lone pairs. These bonds can be any combination of single, double, or triple bonds.
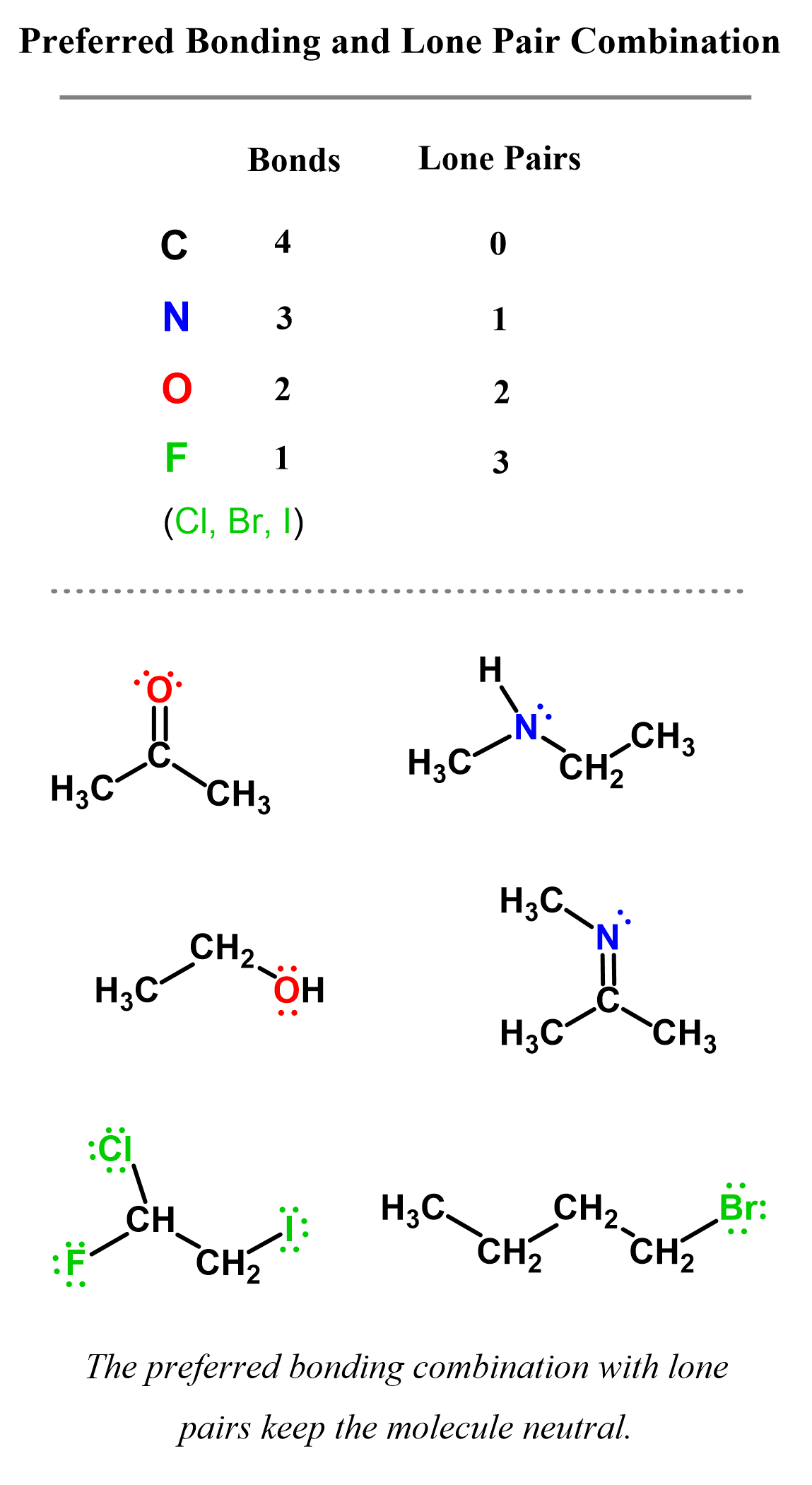
Here are some examples of carbon, nitrogen, oxygen, and halogens in organic molecules where the preferred bonding combination with lone pairs keeps the molecule neutral:
When carbon loses one of the bonds by giving up the electrons, making that bond, it becomes positively charged. We call these species carbocations.
There is a formula, in fact, several of them for calculating the formal charge, but I recommend you practice enough to recognize the correct combination of bonds, lone pairs, and formal charges, as it is going to be a very important part in your organic chemistry class for drawing resonance structures and understanding organic reactions.

Notice that in carbocations, there is a carbon atom that is negatively charged, and that is because, in addition to the three bonds, there is also a lone pair of electrons.
Check out the following articles for more detailed discussion and practice problems on formal charges and bonding patterns in organic chemistry.
- Lewis Structures in Organic Chemistry
- Valency and Formal Charges in Organic Chemistry
- Bonding Patterns in Organic Chemistry
- How to Determine the Number of Lone Pairs
- sp3, sp2, and sp Hybridization in Organic Chemistry with Practice Problems
- How to Quickly Determine The sp3, sp2, and sp Hybridization
- Bond Lengths and Bond Strengths
- VSEPR Theory – Molecular and Electron Geometry of Organic Molecules
- Dipole-dipole, London Dispersion, and Hydrogen Bonding Interactions
- Dipole Moment and Molecular Polarity
- Boiling Point and Melting Point in Organic Chemistry
- Boiling Point and Melting Point Practice Problems
- Solubility of Organic Compounds
- General Chemistry Overview Quiz
- Bond-Line or Skeletal Structures
- Functional Groups in Organic Chemistry with Practice Problems
- Bond-line, Lewis and Condensed Structures with Practice Problems
- Curved Arrows with Practice Problems
- Resonance Structures in Organic Chemistry with Practice Problems
- Rules for Drawing Resonance Structures
- Bonding Patterns in Organic Chemistry
- Major and Minor Resonance Contributor
- Significant Resonance Structures
- How to Choose the More Stable Resonance Structure
- Drawing Complex Patterns in Resonance Structures
- Localized and Delocalized Lone Pairs with Practice Problems
- Molecular Representations Quiz

Is there some moveable magnetic mechanism that could sweep the air above industrial zones, and above large downtown cities, that could remove the dense suspended carbon dioxide in the air by attracting its positive charge?